In the pharmaceutical industry, data integrity plays a pivotal role in maintaining the quality, safety, and
efficacy of their products. Good Manufacturing Practices (GMP) are designed to ensure that all aspects of drug production adhere to regulatory standards, with data integrity being a cornerstone of these practices. This article delves into the importance of data integrity within GMP environments, explores the challenges associated with it, and provides some of the best practices for its maintenance.
Understanding Data Integrity in GMP Context
Data integrity refers to the accuracy, consistency, and reliability of data throughout its lifecycle. In a GMP
environment, this means that all records whether paper-based or electronic must be accurate, complete, and traceable. The integrity of data ensures that decisions made during production and testing are based on reliable information, which is crucial for producing safe and effective drugs.
Principles of Data Integrity, the ALCOA++
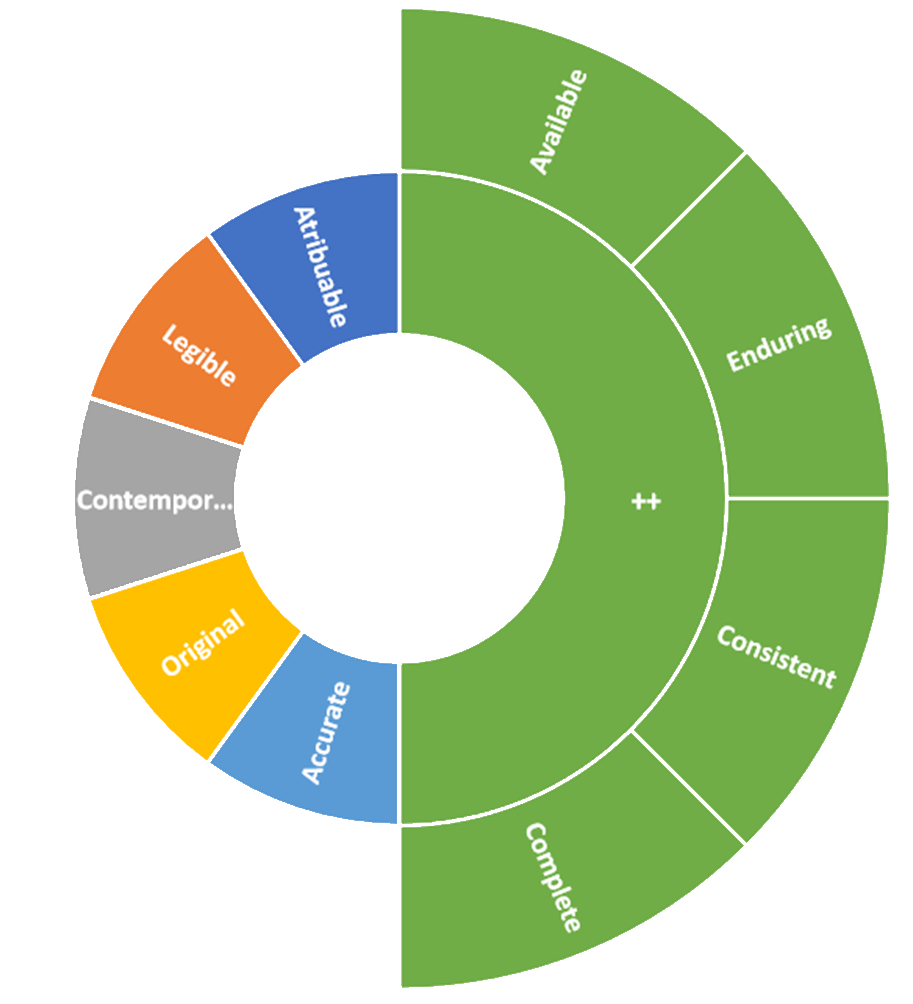
- Accurate: This means the data must be free from errors and correctly reflects the actual observations or measurements.
- Legible: The data should be readable and understandable, both in terms of format and content.
- Consistent: Data should be uniform and follow defined standards to avoid discrepancies.
- Original: It refers to the source of the data being authentic and not altered or falsified.
- Attributable/auditable: The data should be attributable and retrievable when needed, ensuring it’s available for review, audit, or regulatory inspection.
- ++
- Complete: Ensuring all necessary data is captured and nothing is missing.
- Available: Related to accessibility but might emphasize the timeliness of data retrieval.
- Secure: Protecting data from unauthorized access or tampering.
- Traceable: Ability to track the origin and history of data changes (i.e. audit trail).
- Compliant: Adherence to relevant regulatory requirements and standards.
Challenges in Maintaining Data Integrity
- Human Error: Human error remains a significant challenge in maintaining data integrity, particularly due to manual data entry processes. Errors can range from simple typos to misalignment of data fields, which can lead to serious consequences such as incorrect conclusions or dosing errors in pharmaceutical settings. For instance, a misplaced decimal point in dosage instructions for a reagents could result in life-threatening product. Additionally, stress and fatigue among employees can increase the likelihood of mistakes. Implementing thorough training programs and standard operating procedures (SOP) can help mitigate these issues by fostering a culture of careful data handling.
- System Failures: Technological issues such as hardware malfunctions, software glitches, or power outages are common threats to data integrity. A server crash or software bug can lead to data loss or corruption, potentially disrupting research or operations significantly. For example, a sudden power outage in a QC database could erase crucial data, compromising the production or product results. Ensuring robust backup systems and failover mechanisms detailed in a disaster recovery plan is essential to prevent such disruptions.
- Complexity of Data Management: As pharmaceutical processes become more advanced, managing large and diverse datasets from different systems becomes increasingly complex. Integrating different systems, ensuring compliance with regulations, and handling the volume and variety of data can all challenge efforts to maintain integrity. For example, combining data from multiple sources for a certificate of conformities/authenticities requires meticulous management to avoid inconsistencies and a solid Quality Assurance departement to prevent such inconsistencies into the steps of the product release.
- Cybersecurity Threats: The increasing reliance on electronic systems exposes organizations to various cyber threats, including malware, phishing, and ransomware attacks. These breaches can lead to unauthorized access or alteration of sensitive data, undermining trust with stakeholders. For instance, in a worst case sceanario, a breach in a pharmaceutical company’s database could expose confidential patient information, leading to legal and reputational damage. But “smaller” thread, like malware, phishing, and ransomware attacks may impact or block the production. Implementing encryption technologies, training peoples, conducting regular security audits, and implementing basics IT practise like segmenting the network are critical defenses against these threats.
Best Practices for Ensuring Data Integrity
- Implement Robust Quality Systems: Establishing a comprehensive quality management system (QMS) that includes detailed SOPs for every aspect of data handling, ensuring consistency and accountability throughout the data lifecycle.
- Use Validated Computer Systems: Ensure software systems undergo thorough validation processes, including Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ), to confirm they function as intended and remain reliable over time.
- Training Personnel: Ensuring that staff are well-trained in GMP practices, related to data handling and specific software systems used, ensuring staff are well-prepared to handle data accurately and securely.
- Regular Audits, periodical reviews: Conducting routine audits and periodical reviews helps identify potential gaps or issues early, allowing for timely corrective actions.
- Data Backup and Archiving: Develop reliable backup systems with scheduled backups (e.g., daily) and establish retention policies for archived data, ensuring redundancy to mitigate risks of data loss.
- Ensure Continuous Monitoring and Role-Based Access: Continuously monitor computer systems for performance issues and implement role-based access controls to restrict data modification to authorized personnel only.
- Maintain Data Consistency Across Platforms: Ensure data consistency by maintaining records across different platforms and systems.
- Minimize Human Error: Implement automated tools with error-checking features and establish a system of checks and balances, such as multiple reviews before data finalization, to minimize errors.
- Maintain Detailed Documentation for Traceability: Keep thorough records of all data handling processes to facilitate audits and ensure traceability, balancing detail with manageability for staff.
- Adapt to Evolving Technologies and Standards: Regularly review and update data integrity policies to align with current and new regulatory, ensuring ongoing compliance and effectiveness. Incorporate relevant frameworks such as ISO/IEC 27001, ISA/IEC 62443, etc..
The Impact of Non-Compliance with Data Integrity Standards
Non-compliance with data integrity standards poses significant risks to pharmaceutical companies, impacting various facets of their operations and reputation. Regulatory bodies impose strict guidelines to ensure data accuracy and reliability, and failing to meet these standards can result in legal actions and substantial fines. Furthermore, compromised data integrity can lead to the production of substandard drugs, endangering patient health and safety.
The repercussions extend beyond regulatory issues; they affect a company’s financial stability, as non-compliance can trigger costly product recalls and lawsuits. Reputational damage is another critical concern, as trust is vital in the pharmaceutical industry. Loss of customer loyalty and partnerships can hinder business growth and success.
Operational disruptions are also a consequence, often resulting from issues like equipment malfunctions or data integrity issues. These disruptions lead to production halts, which not only result in lost revenue but also delay the delivery of life-saving drugs to market, potentially worsening health outcomes and straining healthcare systems. Beyond immediate financial losses, such disruptions can cause supply chain issues, harm a company’s reputation, increase operational costs due to repairs and maintenance, and affect employee morale and retention. Additionally, production halts can reduce funds available for research and development, slowing innovation and growth. Overall, operational disruptions have far-reaching consequences, impacting a company’s financial stability, market position, and ability to fulfill its mission in improving public health.
Difference Between GMP Data and Non-GMP Data
In the context of data integrity, understanding the distinction between GMP (Good Manufacturing Practices) data and non-GMP data is crucial. Here’s a breakdown:
GMP Data:
- Definition: GMP data refers to information related to manufacturing processes, raw materials, finished products, and activities that directly impact product quality and safety.
- Key Characteristics:
- Must adhere to strict regulatory guidelines to ensure product consistency and compliance.
- Requires rigorous documentation, accuracy, consistency, and security throughout its lifecycle.
- Examples include batch production records, raw material certificates of analysis, equipment maintenance logs, and laboratory test results.
Non-GMP Data:
- Definition: Non-GMP data pertains to information that does not directly influence product quality or
safety. - Key Characteristics:
- Subject to less stringent controls as its impact on product quality is minimal or nonexistent.
- Typically used for operational efficiency, process optimization, or general business management.
- Examples include employee training records, meeting minutes, and general office memos.
Distinction:
- The primary difference lies in the purpose and use of the data. GMP data is essential for ensuring regulatory compliance, product quality, and traceability, whereas non-GMP data serves other business purposes without direct ties to manufacturing processes.
- While some administrative tasks may indirectly affect GMP compliance (e.g., equipment maintenance schedules), their classification depends on their impact on product quality.
Conclusion
Data integrity is essential for ensuring that pharmaceutical products meet the highest standards of quality and safety. In a GMP environment, maintaining data integrity requires a combination of robust systems, rigorous training, and continuous monitoring. By addressing challenges head-on and adhering to best practices, companies can safeguard their operations, protect patient welfare, and uphold regulatory compliance.
Ultimately, a commitment to data integrity is not just a regulatory requirement but a fundamental aspect of corporate responsibility in the pharmaceutical sector.



